UCI Student Researcher Hopes Super Polymer Will Change the World
By Jonathan Cheung
08.09.05 -- Imagine a concrete wall that can
|
absorb blow after blow from a sledgehammer, remaining unbroken no matter how much force is applied. Or an airplane, made of an incredibly light material that will not rupture in the event of a crash. These super materials may be the stuff of science fiction, but the modules that make them possible could be available for commercial use in as few as five years, according to UCI chemistry graduate student Aaron Kushner.
“Basically, I am making a high-tech polymer that shatters the rules of today’s synthetic polymers,” Kushner says. “The superstructure of my polymer incorporates another level of order in the form of tiny, specifically designed modules built into the main polymer backbone.”
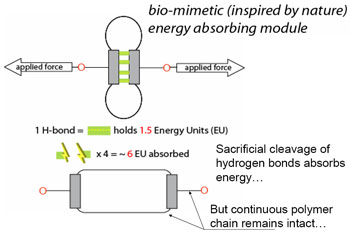
The modules are held together by four hydrogen bonds and two covalent bonds. Each covalent bond forms a folded loop, which takes much more energy to break than the four hydrogen bonds combined. When force is applied, one module’s hydrogen bonds break and the module elongates with its covalent bonds intact, absorbing the energy. If more force is applied, the next module’s hydrogen bonds cleave before the first module’s covalent bonds are broken. As the force increases, the material remains undamaged because each module’s hydrogen bonds break sequentially and leave the covalent bonds unbroken. When the applied force is removed, the unfolded modules resume their state of lowest energy and the hydrogen bonds reform, ready to break at the next assault.
|
“Today’s polymers are random lines that get stretched and can break very easily,” adds Vahe Gabuchian, a third-year mechanical engineering and chemistry double major and SURF-IT research fellow. “These modules are little units; when force is applied, the units’ internal hydrogen bonds break, but the chain between the units remains intact.”
The result is a true elastomer: a flexible, strong material that is able to support a large amount of weight and yield to great force without breaking.
The modules can also be added to existing materials to provide extra support.
“Take any kind of polymer material that is used for any structural or engineering function, add some of this module and it suddenly becomes unbreakable,” Kushner explains.
|
He points to nature as the original inspiration for the design. A few years ago, Zhibin Guan, associate professor of chemistry and Kushner’s faculty mentor, began studying titin, a muscle protein that uses the technique of modular unfolding domains to maintain perfect elasticity. Guan, a polymer chemist, adopted the concept, mimicking biological techniques and incorporating them into polymers.
Currently, Kushner is synthesizing two different versions of the module, which is only the first step in the project. Next, he will implement a testing phase to observe how loop size affects the material properties and how the bulk materials differ from non-module-type polymers. When testing is complete, the module may change the world as we know it.
“The modular polymers that combine mechanical strength, toughness and elasticity potentially can find wide applications,” Guan articulates. “These range from biomedical implants, such as heart implants and spinal disk implants, to defense applications, such as protective apparel.”
And possibly, unbreakable walls and indestructible planes.